[Image: from CatSalut website]
A slide on the future of HIT, from the openEHR conference hosted by the Catalan Health System (CatSalut), 06 June 2023, Barcelona.
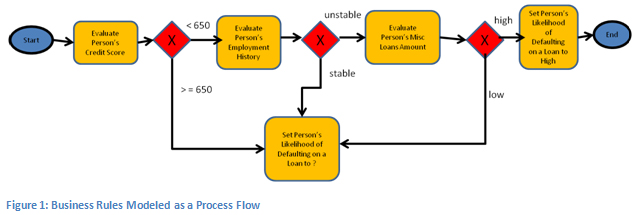
WHAT
- knowledge-based – computational representation of foundational knowledge: ontologies, terminology
- model-based – computational representation of operational knowledge: information and process definitions
- process-based – patient care pathway as first order computational entity: derived from local or published computable guidelines
HOW
- Take all hidden semantics out of the software and DB schemas and represent them as first-order entities, created by domain experts, not IT people
- Realised in a services-based open platform, based on terminology, models, model-driven software, and care pathway execution
- Used to create a system for representing and tracking care pathways, and at each task and decision point, we have a transparent user/computer interaction – not a pile of hidden ‘business logic’
- Voice interaction – voice + models allows for constrained vocabularies (efficient for voice) and goal-oriented navigation rather (user-driven) not form-based navigation (developer-driven); documentation is created ‘on the way’
- Machine learning – use of AI created via supervised training of blank LLMs to perform patient-specific reasoning on the data
RESULT
Signs of success:
- Engineering: we get rid of applications – evolve to ‘task-oriented IT’
- Administrative: we get rid of ‘referrals’ – evolve to ‘straight-through care’
- Clinical: single-source-of-truth medications list, no more ‘med rec’ – evolve from institutional copies to a true ‘digital twin’
- Patient records: we get rid of separate clinical documenting – ‘documenting while doing’
- => a patient-centric care experience.